
High content efficacy data
Discovering the impact of a potential drug on disease model’s physiological parameters is the base of an efficacy characterization study. Indeed, physiological parameters are used to track the evolution of a disease and any change can be proof of its improvement or worsening. At SYNCROSOME, we consider recording physiological parameters as an obligation in our studies. Hence, some parameters are common for all our models (e.g.: recording the weight, evaluating specific behavior corresponding to pain or unease). For organs physiological analysis, surgeries are performed in our dedicated surgical room. On the other hand, our scientists have developed specific techniques and have mastered specific technologies to obtain the most relevant data for evaluating a molecule efficacy.
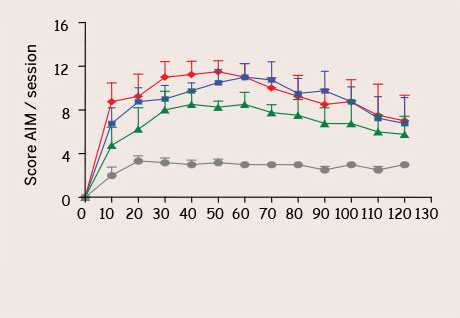
With behavioral tests such as stepping test, initiation test, cylinder test or bar test our Central Nervous System (CNS) department evaluates motor and locomotor activity. Specific parameters like AIM (Abnormal Involuntary Movements) or scores like MNSS (Modified Neurological Severity Score) – which evaluates motor and sensorial activity, equilibrium and reflexes – can also be recorded.
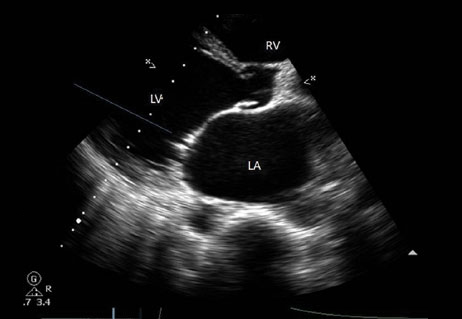
Our Cardiology (CV) department monitors parameters such as Blood pressure, heart rate, mean arterial pressure, blood volume, right ventricular pressure… These parameters can be measured punctually by classical catheterization or continuously by telemetry. We also have the capacity to characterize infarct size by planimetry and to evaluate vascular micro constriction by Doppler echocardiography.
Several physiological read-outs are recorded routinely by our Gastroenterology (GI) department. Some parameters such as the number and intensity of abdominal contraction or retching, vomiting and nausea-like behavior are evaluated following a specific protocol. Other records like DAI (Disease Activity Index), colon length and weight or gastric lesions are measured and quantified. We also have the capacity to perform anorectal manometry.
Depending on the disease model, our Respiratory (RESPI) department records symptomatic read out such as whole body plethysmography (tidal volume, respiratory rate, expiration time, respiratory flows, PenH, etc…). After surgery, the ratio lungs/body weight can also be calculated.
Standard analysis like Glycemia, Insulinemia, Glucose Tolerance Test (GTT) or Insulinoresistance (HOMA Index) are performed by our Metabolic Diseases department on their animal models of diabetes.
For a better characterization of your molecule’s impact on a pathology we have mastered several methods of functional exploration. Thus we measure parameters repeatedly or dynamically during operation of an organ or group of organs working in synergy. Echocardiography is used to evaluate morphological and functional parameters of the heart and blood vessels in small laboratory animals. It is a non-invasive technic that can be performed on different species (mouse, rat, rabbit…). Associated with a Laser Doppler it is interesting to evaluate vascular micro constriction. We also have the capacity to characterize an infarct size by planimetry. We use plethysmography for measuring blood pressure in awaken mice or rats. This non-invasive method can be repeated as many times as necessary during an animal’s life.
For specific studies we also have individual metabolic cages allowing controlling the food intake (food and drink) and collecting separately excreted substances (urine, faeces). Metabolic controls are used to study various functions such as feeding behavior, renal function, digestive function…
To have a better insight of functional behavior on a cellular level, we perform microscopic evaluation of an organ or group of organs working in synergy. After collections, organs are prepared then sliced with a cryostat for labeling on microscope slides. For the analysis of these labeled tissues, SYNCROSOME has access to a cell-imaging platform (PICSL) where we can perform confocal microscopy, photon microscopy, video-microscopy…
Functional Exploration
Cellular, Molecular and Inflammatory Biomarkers for preclinical diseases
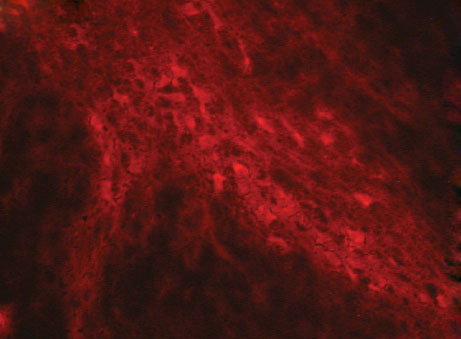
In order to improve a molecule’s efficacy evaluation, SYNCROSOME has developed several tools to record parameters related to Cellular and Molecular Biomarkers for preclinical or animal disease models. This histological approach is a part of our will to perform in depth efficacy characterization. These preclinical biomarkers give a better understanding of your candidate drug impact on a microscopic level. Our scientific team, who has mastered cellular and molecular labeling protocols, has also developed ImmunoHistoChemistry (IHC) protocols dedicated to specific disease models. For example, our CNS department evaluates the impact of a neurological compound on neuro-protection and neuro–inflammation by monitoring neuromediators, cellular counting in specific brain regions or glia or microglia proliferation. Our Respiratory department has developed protocols for counting number of apoptotic cells or eosinophils / neutrophils and for evaluating cells and macrophages infiltration as well as vascular remodeling.
As a large part of our preclinical disease models being based on inflammation preclinical evaluation, we also developed ELISA tests to dose inflammatory molecules and follow the decrease of inflammation (inflammatory preclinical biomarkers).
HPLC analysis allow to dose neurotransmitter such as Dopamine, Glutamate, Serotonin, GABA, Noradrenalin… We also offer HPLC-MS/MS dosage to evaluate your compound’s concentration in blood (and measure parameters such as Cmax, Tmax or AUC) as a part of PK/PD studies. This technology can also be used in our first Proof of Concept (POC) studies, which allow screening several compounds at once.
Whatever molecule or compound you would like to dose, our scientific team will work closely with you for establishing a tailored method dedicated to your needs.
At SYNCROSOME, we are dedicated to characterize a molecule’s efficacy as accurately as possible. Our goal is to offer you high clinical predictability by providing high quality data. We have developed several animal models dedicated to a specific pathologies in our five main therapeutic areas :
- Central Nervous System CNS disease models
- Gastroenterology disease models
- Respiratory disease models
- Cardiovascular disease models
- Metabolic diseases models
Moreover our scientific team’s wide range of skills allows them to perform in vivo experiments answering to your needs. We possess an accredited vivarium and surgery suites and have the capabilities to test different type of administration roads for your compounds.
We can conduct your in vivo Pharmacokinetic (PK) and Central Nervous System-Pharmacokinetic (CNS-PK) studies on rodent and non rodents with reproducible results. Depending on your needs, we are able to provide many sorts of data which can either be sent to you or be analyzed by us.
For your CNS targeting compounds we can perform acute and multiple blood and Cerebro Spinal Fluid (CSF) sampling. With microdialysis (or retro-dialysis for high performance brain administration) we can assess in vivo the Blood Brain Barrier passage of your molecule.
In Vivo Experiments



